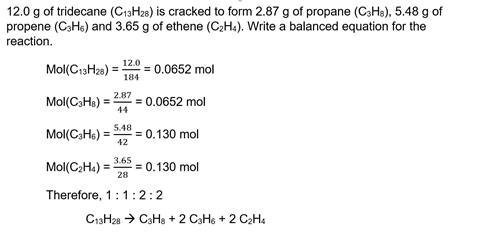
Unbelievable Info About How To Find Out Moles

Number of moles formula is.
How to find out moles. Mass of one mole mno2 = 86.94g. So a mole of water (h2o) has a mass of 18 g. Ask me questions on facebook:
Number of moles = \(\frac {mass\; Avogadro’s number represents a mole ( often abbreviated mol ), and it states that a mole of a substance has (6.022×1023) or 6.022×10^23 molecules per mole or atoms. A stands for asymmetrical shape, which is generally the shape of.
A mole of carbon dioxide. Number of moles = 95. Mole}\) uses of mole concept.
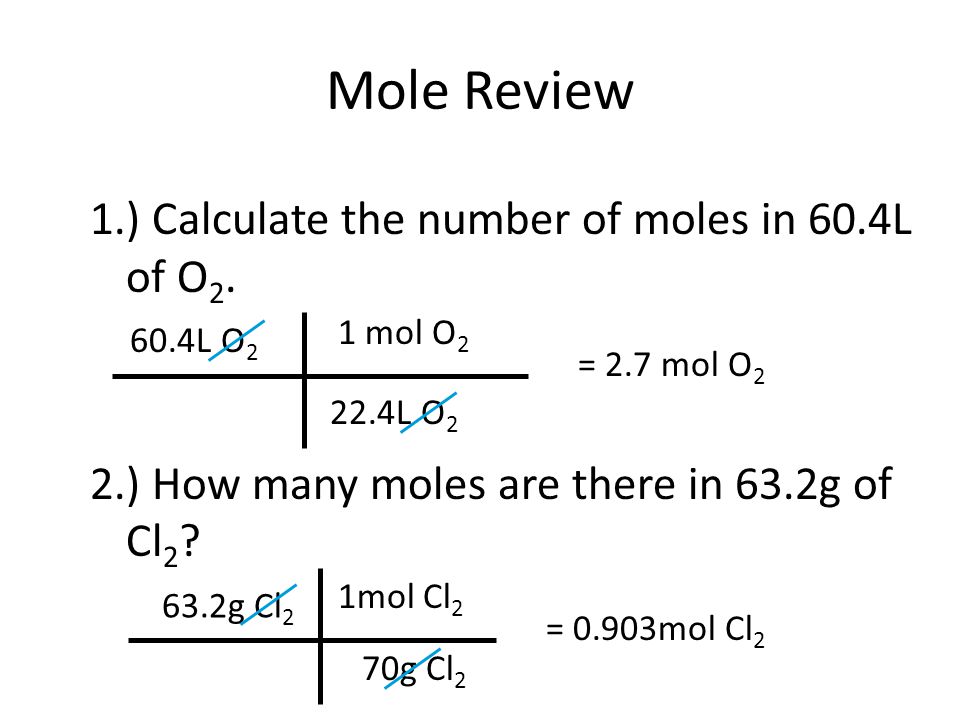
N = m / m where n = number of moles m =. The formula for number of moles. Number of moles = mass of substance / mass of one mole.
Unbalanced chemical reaction step 1:. The number of moles formula is given as follows: Mass of mno2 = 95g.
A mole of a molecular compound contains 6 x 1023 molecules. The given chemical reaction is not balanced since the. In the abcdes of moles, each letter represents the first letter of what to look for while checking your moles.
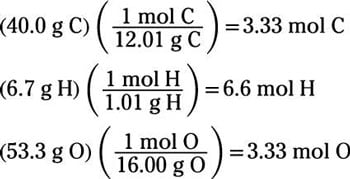


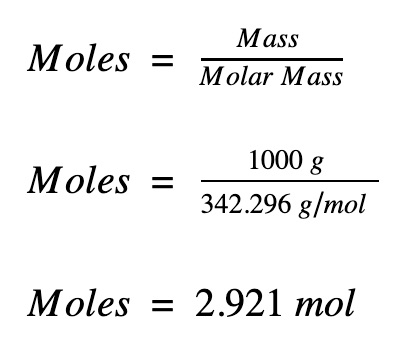

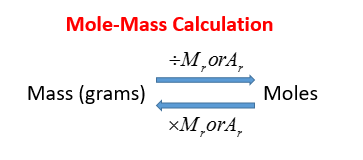





/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)




